Conserving Outdoor Metal Sculpture
Rupert Harris
 |
|
 |
The Quadriga, Wellington Arch, London, a bronze sculpture with an artifical patina which has been retouched and rewaxed (above), and (right) the patina at the start of work |
This country' s public works of art enliven our landscape and stand as testaments to our rich social, cultural and political history. It is essential that those bodies with the responsibility to care for our public sculptures respect their vulnerability and implement sound conservation and maintenance practice. This article provides a brief introduction to the materials and processes involved in the manufacture of external metal sculptures and the problems associated with their care.
Various metals have been used to produce sculpture over the centuries, including bronze, lead, cast iron, zinc and copper electrotype. Each has its own sculptural qualities, but equally varied and just as important are the surface finishes and patinas. Their influence on the aesthetic quality of the work of art is immeasurable, and they are often far more fragile than the body of the sculpture itself.
BRONZE
Although it has always been expensive, bronze is ideal for exterior use due to its comparative resistance to corrosion and its ability to take a fine, aesthetically pleasing finish. After construction in the foundry, the surface of the bronze is further finely worked to disguise the joints and to add further detail. It is then given an artificial patina (under the direction of the sculptor) using a variety of chemicals and, finally, coatings of wax and sometimes lacquers to protect it. Traditionally, the artificial patina chosen would usually have been a dark or midbrown colour, though over the past century other colours and shades have also been used by artists on their bronze castings.
Structural damage to bronze sculptures is comparatively rare and is usually caused by either the failure of patches and joints made when the piece was first cast, failure of the internal armature and fixings, or impact damage (from trees falling or road traffic accidents for example). The aesthetic surface of the bronze is, however, far more vulnerable to damage: if the protective coatings of wax are not maintained they are eventually lost, leaving the original artificial patina unprotected from environmental influence (water, gases and air-borne pollutants). This leads to slow corrosion of the bronze and the development of a natural patina, usually mid to light green in colour.
The corrosion that causes the natural patina is slow to form and usually stable, however the lack of protective coatings can lead to areas of active, aggressive corrosion on the bronze, which can cause permanent damage to the surface.
The green copper sulphates forming a natural patina are washed downwards by rainfall and cause permanent staining to stone plinths. Although the appearance of this staining can be slightly improved by conservation treatments such as chemical poultices, it is almost impossible to remove altogether.
 |
| Active bronze corrosion |
 |
| Copper sulphate staining on a stone plinth caused by rainwater runs |
Lack of maintenance on bronze sculptures is so common that the naturally formed green patina is a familiar sight to us, and is often prized for its own aesthetic value. Generally, when undergoing conservation treatment, the patina, either natural or artificial, should not be removed, but respected as an integral part of the sculpture. However, a natural patina can sometimes include disfiguring marks or black streaks that can make the sculpture difficult to read, and it is generally accepted that these may be removed and the surface manipulated to reinstate a cohesive whole.
If an artificial patina requires conservation or retouching, it is most important to use the same methods and chemicals in their treatment as those used originally. A few modern alternatives are available, but this author has yet to see any new methods in this field which compare favourably to the quality of the result achieved by using the same materials and techniques as were originally used in the foundry during the production of the piece: methods which have changed little from antiquity to the present day for good reason.
LEAD
The production of lead sculpture reached its height in England during the 18th century. At that time it became cheaper to produce cast leads painted to resemble stone than to acquire genuine stone sculpture, so leads were frequently painted with lead-rich oil-based paints to give the appearance of limestone or marble. Many were also painted in naturalistic polychrome. In most cases the original paint will have weathered away, though traces may have survived in recessed areas, and it is essential to take small samples for analysis before proposing conservation treatment, and to record the results of the analysis in order to retain this vital map of the object's aesthetic history (see page 159). Original or historic paint should not be removed and should be left undisturbed as far as possible, even if a curatorial decision is made to restore the sculpture to its creator's original intention and repaint it in its original scheme.
Lead itself is generally resistant to corrosion and, if left exposed, the surface will develop a protective, pale to dark grey patina. As so much lead sculpture has not had its paint surface maintained over the years, we are now culturally used to seeing lead with its natural patina, in the same way that we accept naturally formed green patina on bronzes and lichen on stone. As a result, these finishes have become part of our aesthetic understanding of historic sculpture.
Most lead sculpture was cast in one piece with a core material of sand, plaster, or a ground refractory material such as brick. The iron armature, and usually the core material, were left inside after the casting process was complete to provide support to the soft lead. It is the original armature that is the sculpture's most threatening problem, since it will inevitably corrode. As it does, the rust expands up to ten times the volume of the original iron, causing the lead to split, slump and eventually collapse.
As a soft material, impact damage is often found on lead sculpture. Superficial dents and scratches to the surface are most common, and the material is particularly vulnerable to graffiti and other forms of vandalism (shotgun pellet damage is not unusual). There are even cases where lead sculptures are being gnawed by squirrels.
 |
 |
 |
 |
| The repair of a lead sculpture with a rusting armature: top left and right, the sculpture is opened up to remove the sand core and expose the rusting armature; above, the new stainless steel armature; and, right, lead burning the patch back into place. |
The conservation of lead sculpture therefore often involves extensive structural repair, including removal of the original armature and core material, followed by the insertion of a new stainless steel armature. This will probably require cutting window-like patches to allow access. These are then lead-burned back into place, and the seams carefully worked back and chased using fine chisels and tools to match the original surface. Additionally, if areas of the sculpture have become crushed or collapsed it is sometimes necessary to cut further patches in order to gain access to dress out the lead from the inside. Alternatively, crushed areas may be built up by adding successive layers of lead (by lead burning), then carving back to the original form. These treatments, though intrusive, enable the structural integrity and original sculptural form of the piece to be restored, and if performed by experienced conservators, ensure the sculpture will retain the appropriate, aged appearance and recover its structural stability.
It is generally considered poor practice to remove a natural patina which, usually, will have been acquired by the surface of the lead after many years in the open air. Nevertheless, it may be appropriate to selectively remove intrusive stains if they interfere with the reading of the sculptural form, and indeed a curatorial decision may be made to recreate the original painted surface for other reasons.
If left unpainted, repaired areas on the lead will be visible due to their shinier, worked surface. One solution (preferred by the author) is to disguise these areas with a thin wash of casein-based watercolour paint. As the paint slowly wears off in an outdoor environment, it is replaced by natural patina. Unpainted lead requires little maintenance: monitoring of condition and an annual wash will usually be all that is required.
 |
| Shotgun pellet damage on a lead sculpture which still retains some of its original stone-coloured paint. |
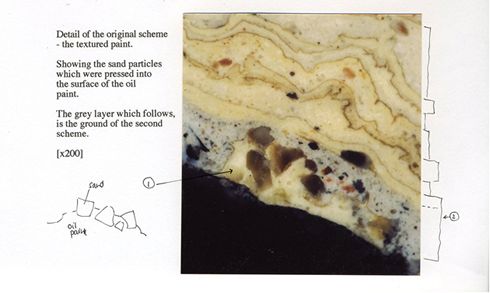 |
| Micrograph analysis of a paint sample taken from 'Fanny's Urn', a small lead urn in Sir John Soane's Museum |
CAST IRON
During the 19th century the development of new technologies and the Victorian enthusiasm for ingenuity of invention in industrial processes lead to new, cost-effective materials being used to cast sculpture, in particular cast iron, zinc and copper electrotyping or electroforming. The majority of this sculpture was intended for exterior display.
Cast iron is a brittle material, making it susceptible to impact damage and stresses caused by expansion rusting of construction joints in the object. Cast iron can be repaired using a range of techniques such as pinning, stitching or welding. However, great care is required when attempting repairs, especially if heat is involved, as in welding, because localised thermal expansion can induce further cracking and subsequent structural failure.
As iron rusts rapidly if unprotected, almost all iron sculptures were originally painted. As with lead sculpture, or indeed any important painted object, it is vital that existing paint is analysed so that original paint schemes can be identified and recorded, as much iron sculpture has been over-painted many times in its life.
Thick layers of paint can obscure sculptural detail, so conservation may involve their careful removal, followed by the application of primer, mid and top coats of paint to protect the surface from moisture. (For further information, see Keith Blackney's articles on cleaning and painting ironwork which are also available on this website.) The colours chosen must be dictated by the results of analysis and where possible other historical research.
 |
| The paint surface of this cast iron sculpture has not been maintained, allowing the iron to rust. |
ZINC
The manufacture of large-scale sculptures in zinc was only in fashion for a short time between 1860 and 1900 and as a result they are relatively uncommon. The sculptures were cast in sections and assembled using lead soldered joints, which were additionally pinned onto structural elements such as iron straps or frames, or zinc plates, for extra strength.
To achieve a bronze effect, the completed sculptures were frequently electroplated in copper, then chemically patinated and protected with layers of varnish or wax. In Britain, the firm of Elkington was instrumental in the development of electroplating technology and this company often finished zinc sculpture which had been imported from Germany, France or America.
Today, zinc sculptures sited outdoors have usually lost their original surface coatings and copper plating through corrosion, exposing the grey-coloured zinc. The corrosion causes a very pitted surface in the zinc, and this is usually accompanied by failure of the lead solder joints. The result compromises the object's structural stability and leaves it vulnerable to serious fracture as well as ongoing corrosion processes, further leading to the rarity of sculpture in this metal. Structural repairs can be effected and the corroded surface restored, if appropriate, to replicate the original 'bronzed' appearance.
 |
|
| Zinc corrosion causes pustules to form which, when cleaned, reveal deep pits in the surface |
ELECTROTYPES
The process of electrotyping, or 'electroforming', was developed in the 1830s. There were two methods of manufacture, the method chosen depended on what was being made as the technique was employed for many different kinds of objects and works of art, from small furniture trimmings, tabletop sculpture and relief plaques, to life-size sculptures intended for outdoor display. Usually, smaller objects were made using a wax or plaster model (that is to say, a positive) possibly with an iron armature already inserted for support. Large objects and sculpture were usually made in sections using a number of moulds (that is to say, negatives). The model or mould was placed in a copper-plating tank and copper was allowed to deposit itself by electro-deposition onto the surface of the positive object or negative mould, until it had built up to an adequate structural thickness.
In the case of the model option, the copper built up on the outer surface of the object, so the thicker the copper became, the more surface detail was lost. It was therefore not a good technique for large objects because you could not gain sufficient thickness of copper to retain much strength without losing visual quality. To counteract the potential weakness of this method, the wax impregnated plaster model would often be left inside the object to give support.
In the case of a mould, however, the copper built up from the outer surface inwards, so a greater thickness could be built up without blurring the visible surface of the sculpture, thus making it suitable for large objects where a greater thickness was required for structural strength. This process produced highly detailed surfaces, reproducing every mark of the sculptor's hand.
 |
 |
|
| Water ingress causes electrotype objects to split. Repairs (above right) were carried out using copper sheet. | ||
When a large object was made by the mould method, each copper section was then cleaned and joined together by brazing or soldering. The thickness of the copper that could be deposited meant that iron armatures were seldom required. Electrotypes can be patinated in the same manner as bronzes, but now frequently have a natural, green patina which has developed on the copper over time. Either way, they are easily mistaken for bronzes at first glance.
Those electrotype sculptures which have a very thin copper shell are particularly susceptible to corrosion if not cared for. Where constructed with a plaster core and possibly an iron armature, any consequent ingress of moisture results in the expansion of these materials, leading to splits and eventual collapse. Repairs can be made by reproducing any large, missing or heavily damaged areas using copper sheet, and weak areas may be reinforced by introducing fibreglass resin. Like lead sculpture, such objects usually require the replacement of any iron armature with new stainless steel armatures and structural fixings.
As with bronze, protective coatings of wax or lacquer need to be applied to protect and maintain the delicate surface.
ESTABLISHING A CONSERVATION AND MAINTENANCE PROGRAMME
Metal sculpture can well survive the rigours of outdoor life if structural damage or aggressive corrosion are identified at an early stage and corrected by a skilled conservator, and if regular and correct attention is given to the maintenance of its protective coatings. Because historically most owners or bodies responsible for the sculpture have largely failed to carry out this comparatively simple requirement, our sculptures are now in varying states of repair and much public sculpture in Britain is in need of urgent conservation treatment.
The conservation of the structure and surface of a sculpture is a specialist practice. It is vital that those entrusted with the care of such objects understand the principles behind good conservation and maintenance and obtain the best advice from experienced conservators before embarking on a program of conservation treatments or establishing a maintenance regime. Work must be specified and carried out by qualified conservators who are sufficiently experienced to have gained the skills required. These include an aesthetic sensitivity to sculptural form, an understanding of the subtlety of surface finishes and knowledge of art history. Verifiable proof of the experience and professionalism of the conservator should always be obtained before commissioning advice or work.
Once a sculpture has undergone full conservation and is put in good order, it is equally important that it is maintained regularly as a matter of course. The goal of a preventive maintenance programme for sculpture is to preserve as far as possible the artist's original intention, while being respectful of the historical experience the object may have undergone (including not only the acquisition of a sound, natural patina but also, in some cases, the damage associated with an historical event, such as shrapnel damage for example). This goal can be achieved by slowing down corrosion, maintaining structural soundness and maintaining or improving the aesthetic appearance, while ensuring the unique and visually varied character of a sculpture's surface is also protected.
In recent years the United Kingdom has seen a marked increase in the commissioning of new public sculpture: works of both traditional and modern materials and surface finishes. Those commissioning each new piece must recognise the ongoing responsibility to annually maintain it, and be mindful of the ongoing budget this requires. They must then actively implement good policy. Conservation usually involves significant cost and disruption: regular maintenance on the other hand is cost effective and, most importantly, protects the sculpture and its aesthetic and financial value.



